Why hydrogen?
First, let us have a closer look at the chemical element hydrogen itself. Hydrogen is represented by the letter ‘H’ in the periodic table and is a colorless, odorless and non-toxic gas. It is the simplest, lightest and most abundant of elements. It does not occur naturally by itself but it can be found almost everywhere on earth in combination with other elements. For example, in combination with oxygen, forming water (H2O) or with carbon, forming methane (CH4).
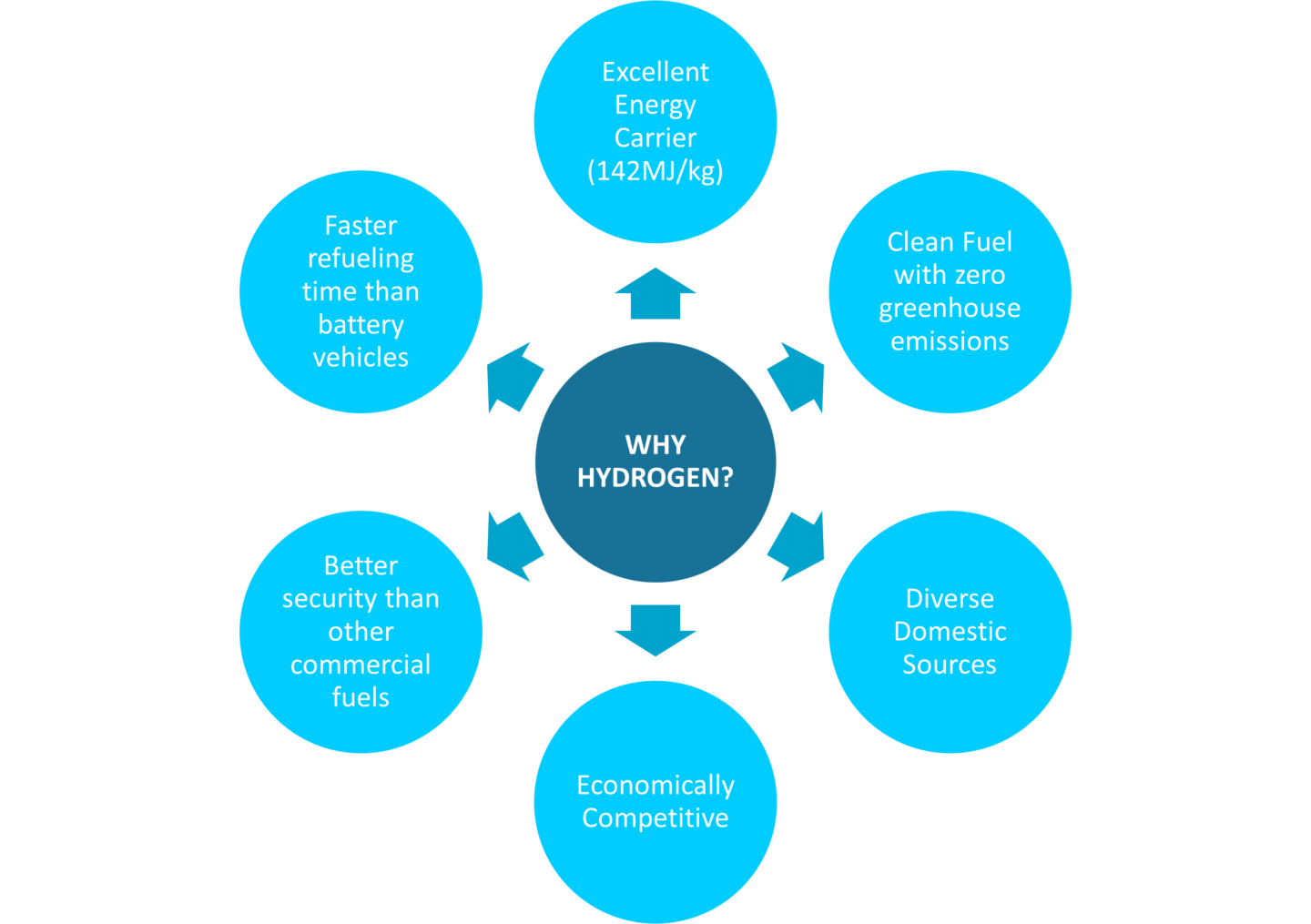
The reason hydrogen is considered as one of the main alternative power sources for the future, is the fact that it has a higher energy content by weight than any energy source we use today [1]. However, since hydrogen has to be extracted (by the process of steam reforming or by electrolysis) from other elements, it is not considered an energy source by itself. It is considered an energy carrier. Energy sources include fossil fuels like coal and oil or renewable energy like wind or water power. Depending on the energy source used for extraction, hydrogen energy qualifies as renewable energy and can be produced with little or no environmental impact (we’ll go deeper into this later in the article). The energy stored in hydrogen can be converted into electrical power by putting it into a fuel cell. The conversion is similar to reversed electrolysis (which is also explained later). These fuel cells can be used to power a wide range of devices, from long distance trucks to small portable laptops. A hydrogen fuel cell does not emit any greenhouse gases, as water and heat are the only by-products.
The high energy content and the low environmental impact are the main reasons to consider the potential of hydrogen in our energy market.
The hydrogen challenge
The reason hydrogen is not yet a key player in our energy landscape is simple: it is difficult to produce, store and transport. As said, Hydrogen is not found on its own and it requires an intensive process to extract the element. It also requires specific infrastructure to be stored and transported.
Production process
Hydrogen is a necessary component in some big industries like chemical production (e.g., making fine oil), metal refining, food processing, etc. These industries make hydrogen using steam reforming, a mature production process in which high-temperature steam (700°C–1,000°C) is used to produce hydrogen from a methane source, such as natural gas, diesel, gasified coal, gasified biomass or a renewable liquid fuel.
Depending on which base resource is used, there is a difference in carbon emissions during production and the hydrogen itself gets another name (e.g., brown or black H2 from coal, grey H2 from gas). Regardless of which source is used, steam reforming will always lead to carbon emissions. Approximately 7kg of carbon dioxide is released for the production of 1kg hydrogen via steam reforming [2]. The process of steam reforming also requires a lot of heat and is highly inefficient (grey hydrogen produced by steam reforming contains less energy than the natural gas which was used as the base resource) [3]. Large industries often use the process of steam reforming to produce their hydrogen on site, as they already have access to steam and heat as a by-product from other industrial processes. This increases cost-efficiency while also tackling the transportation costs at the same time.
Electrolysis
There is another way to produce hydrogen: electrolysis (a.k.a. electrochemical water splitting). This process splits water into its basic elements (oxygen and hydrogen) by using an electric current. The electricity needed for this process can be accessed via renewable sources. You lose 30% of the energy from (renewable) electricity to end up with hydrogen via electrolysis. This loss is bigger than using steam reforming. Offsetting this energy loss is the fact that by using only renewable electricity in the electrolysis process, hydrogen energy can be produced completely environmentally friendly. New types of electrolysis are being developed that will eventually prove to be more energy efficient and provide an emission-free source of energy [4].
Fuel cells
Using a fuel cell to transform hydrogen energy back into electrical energy eliminates any harmful emissions created during this step. It allows for the production and usage of hydrogen energy to be a completely zero emission end-to-end process.
The huge production effort, the loss of energy during the end-to-end production and the lack of easily accessible renewable energy, make the production process one of the main barriers for the widespread use of hydrogen.
Storage & transport
To be able to use and transport hydrogen safely, it needs to be stored efficiently. This can be done in a gaseous or liquid state. Storage of hydrogen as a gas typically requires high-pressure (400–770 bar). Storage of hydrogen as a liquid requires cryogenic temperatures because the boiling point of hydrogen at one atmosphere pressure is −252.8°C. The advantage of liquification is that a liquid hydrogen tank is much lighter than a gasified one. However, the process to liquify hydrogen is more energy intensive compared to the gasified option (+- 40% energy loss to liquify, compared to +/- 13% energy loss for storing hydrogen as a gas). Nevertheless, if our aim is to produce hydrogen completely emission free, it’s more efficient to centralize the production where renewable electricity is readily available and accept the transportation costs and effort.
Domestic hydrogen
The use of hydrogen is no longer limited to big industries. In 2019 the European Commission gave birth to the THyGA project (Testing Hydrogen Admixtures for Gas Appliances). Gas.be is a Belgian knowledge and communication platform that is involved in the THyGA project. They are researching the possibilities of mixing hydrogen and natural gas in our Belgian gas infrastructure. In the mean time, they monitor the impact of this switch on currently installed (and yet to be installed) devices. They do this by verifying safety aspects as well as environmental and energy performance of both domestic and commercial infrastructure and devices [5].
In the meantime, we will already be able to reduce our ecological footprint by using a mixture of natural gas and hydrogen. That is, if we use green hydrogen (hydrogen produced with renewable electricity) [6]. Today, green hydrogen in Belgium is produced on a small scale with excess electricity from our windmills. Our production capacity would allow for a mixture containing up to 30% hydrogen (70% natural gas and 30% hydrogen).
Hydrogen in transport
Besides using hydrogen in our day-to-day households, it also has a big potential in the transport industry. Electric vehicles struggle with range. Adding larger batteries increases the vehicle’s weight which in turn makes it require more energy to move. Hydrogen fueled vehicles have 230 times the energy for the same weight of ‘battery’ (278 Wh/kg (Lithium-ion) to 40.000 wh/kg).
If we can roll out a grid of hydrogen charging points, the future of transportation can be changed forever. Switching to hydrogen from fossil fuels, and even electricity, for private, public and commercial transportation would drastically reduce our carbon footprint without sacrificing any of our current mobility.
Tackling the hydrogen hurdles
As governments worldwide put pressure on energy firms to develop their renewable energy capacities, green hydrogen is looking increasingly attractive. To alleviate some of the current issues in the use of hydrogen, governments have also started to invest heavily in green hydrogen themselves. Finland wants to be a world leader when it comes to hydrogen [7]. And New Zealand declared they want to become the world’s first large green hydrogen producer [8]. The Chilean government recently disclosed plans to invest in green hydrogen production and this in cooperation with the Belgian Port of Antwerp-Bruges. Their idea is to make a green hydrogen hub in the Antwerp Port area, facilitating other companies and industries to use green hydrogen [9][10].
The increasing technological improvements and investments will help hydrogen to overcome its challenges. Hydrogen will definitely play a vital role in the worldwide energy transition and will become a key player in our energy market of the future.
[1] Hydrogen Oil: Green vs Blue, What’s the Difference?
[2] Mitigating Carbon Dioxide Impact of Industrial Steam Methane Reformers
[3] Office of Energy Efficiency
[4] Hydrogen Production: Electrolysis
[5] THyGA | Testing Hydrogen admixture for Gas Applications (thyga-project.eu)
[6] Home – Gas
[7] Hoe Finland wereldleider wil worden op gebied van waterstof
[8] NZ to be the world’s first, largest green hydrogen producer
[9] Port of Antwerp and Zeebrugge partner with Chile on green hydrogen production
[10] Amerikanen pompen 300 miljoen in Antwerpse waterstoffabriek






